A hidden weakness in pancreatic cancer cells
Cancer drugs often target a molecule that cancer cells rely on for survival but, crucially, healthy cells do not. When they are first identified, this type of molecule is known as a therapeutic vulnerability—and what previously made the cancer cell especially fit now makes it an easier target.
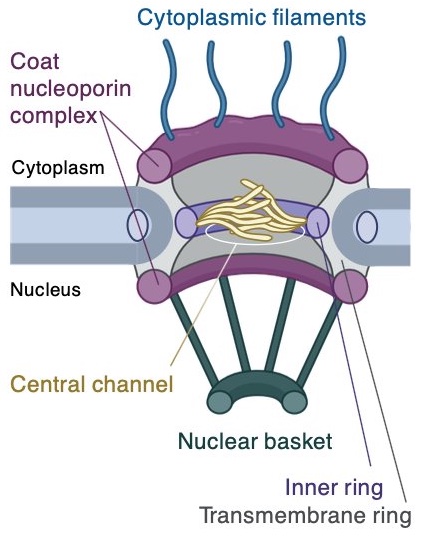
Recently, Damon Runyon Clinical Investigator Steven M. Corsello, MD, and his colleagues at Stanford University School of Medicine identified a new such vulnerability. To build proteins, cells transcribe DNA into messenger RNA (mRNA), which then leave the nucleus and enter the cytoplasm through a tightly controlled passageway called the nuclear pore complex. Because cancer cells grow and divide faster than healthy cells, they need to produce mRNAs at a higher rate, making them especially dependent on the nuclear pore complex to usher these molecules into the cell. In other words, when the nuclear pore is disrupted, healthy cells can recover, but cancer cells cannot.
“Cancer cells are addicted to a constant stream of short-lived pro-survival mRNAs,” Dr. Corsello explained on X. “Starving the cell of key survival signals results in rapid cell death.”
By systematically testing candidates against pancreatic cancer cells, Dr. Corsello and his team found a drug that would do just that. The drug, a previously discontinued cancer drug known as PRLX-93936, functions as a “molecular glue,” recruiting a protein-degrading enzyme to degrade the nuclear pore. In patient-derived pancreatic tumor models, the team showed, this drug effectively induced cancer cell death.
“To date, the insufficient number of unique molecular targets amenable to intervention has been a critical barrier to improved pancreatic cancer treatments,” Dr. Corsello said. “We are excited by the translational potential of this discovery.”
This research was published in Cancer Discovery.
